FDA approved in December of 2016, Restylane Refyne and Restylane Defyne are new Hyaluronic Acid (HA) based fillers to soften and plump lines of expression, such as nasolabial folds (lines from the sides of your nose that run down around the corners of your mouth), marionette lines (the lines running down from the corners of your mouth to the chin), and other lines of expression. Utilizing XpresHAn technology, Restylane Refyne and Defyne are meant to give natural-looking results that will not bunch or form a “shelf” while smiling or expressing. While these fillers are new to us in the USA, Refyne and Defyne were first approved in Europe in 2010 under the name Emervel, showing a proven safety profile with over 1 million treatments worldwide.
XpresHAn technology prolongs the longevity of the filler and allows flex within the skin so that your expressions look natural and smooth. The flexibility of the filler is achieved through a unique manufacturing process, creating additional crosslinking between each HA particle. Restylane Refyne is designed to be very flexible and provide subtle support, while Restylane Defyne is designed to be less flexible and provide additional support. Both products are FDA approved for up to 12 months of longevity.
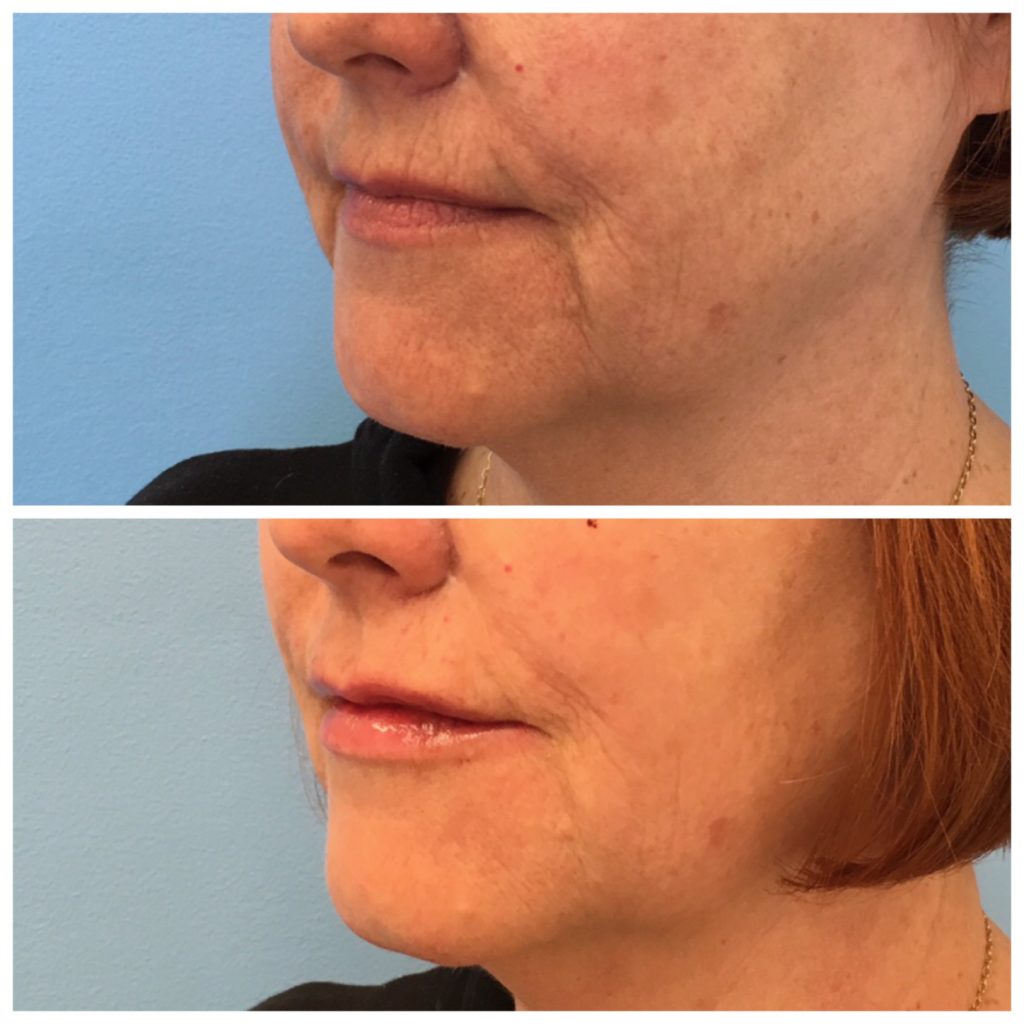
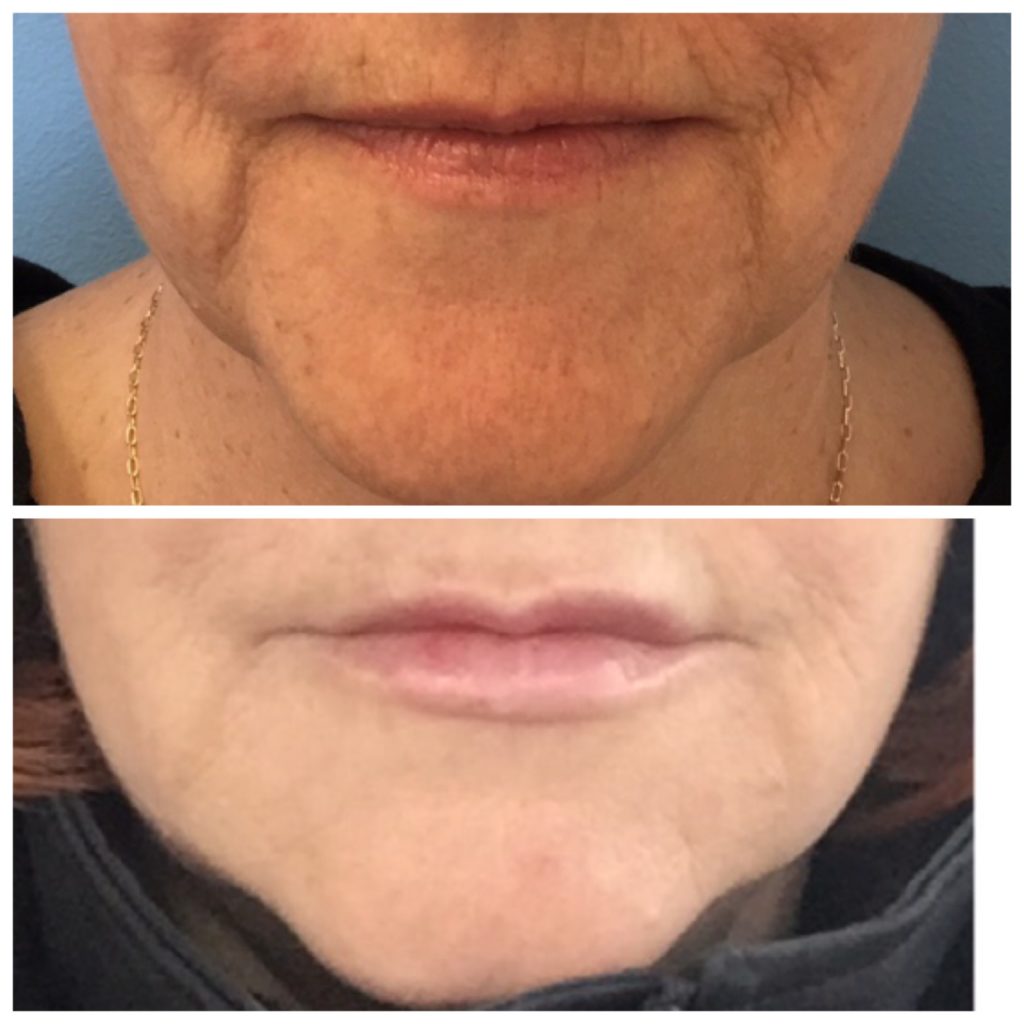
The above images show Restylane Defyne used in the marionettes (the lines that extend from the corners of her mouth to her chin). This product is unlike any other product I have placed into the skin. Once placed, the product is very malleable, leaving patients more satisfied than with other products they can feel in their skin.
What to expect the day of treatment
At the Whole Beauty Institute, we will review your medical history with you and determine if you are a candidate for this next generation of HA fillers. You should not use products within the Restylane family if:
- You have severe allergies with a history of severe reactions (anaphylaxis)
- You are allergic to lidocaine or to any of the gram-positive bacterial proteins used to make hyaluronic acid
You may return to work or your normal daily activity (but not exercise). You might experience some bruising and swelling which is easily concealed with makeup about an hour after injection.
Within two weeks of injection, any bruising and or swelling will have subsided and you will see your beautiful and natural results.
G. (2016, December 17). Galderma Announces FDA Approval of Restylane® Refyne and Restylane® Defyne Dermal Fillers for Treatment of “Laugh Lines”. Retrieved April 11, 2017, from https://www.galderma.com/News/articleType/ArticleView/articleId/120/Galderma-Announces-FDA-Approval-of-Restylane-Refyne-and-Restylane-Defyne-Dermal-Fillers-for-Treatment-of-Laugh-Lines